|
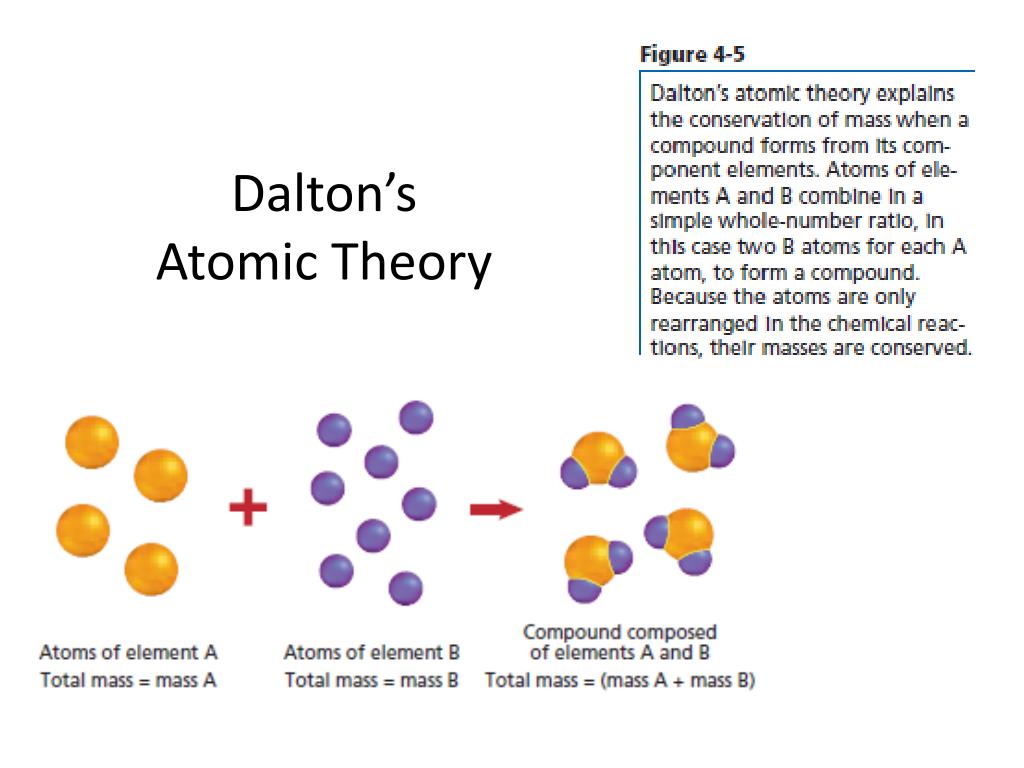
In modern chemistry, Dalton’s atomic theory has been modified due to some of the drawbacks in the principle. 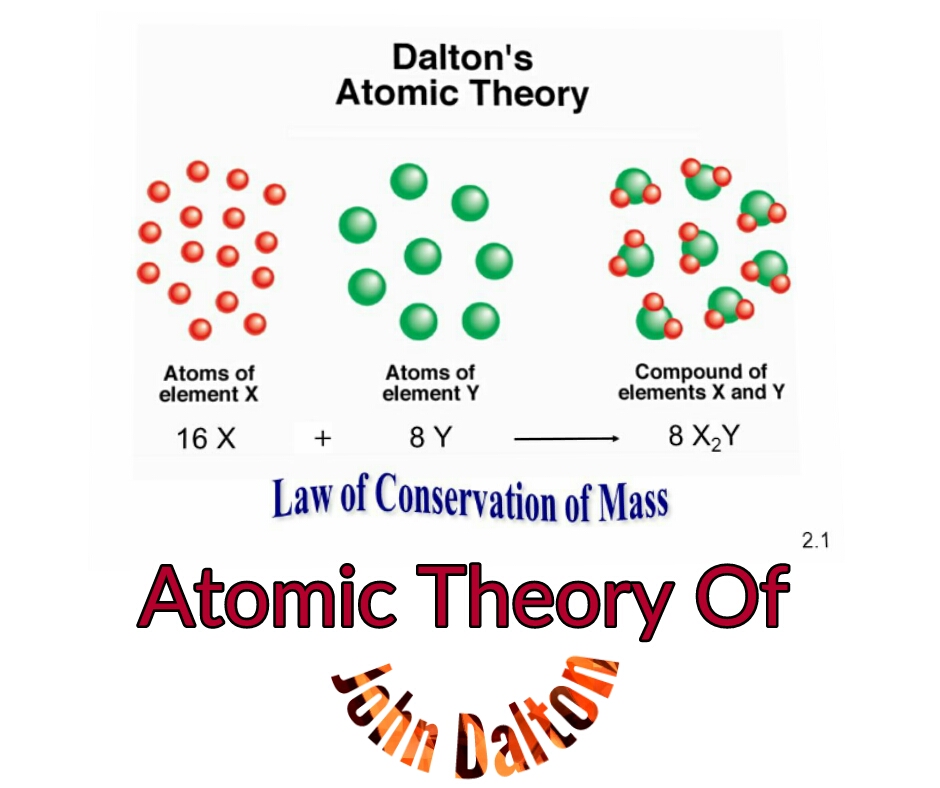
Dalton, in the atomic theory, also introduced the word atom as the most fundamental particle of a chemical substance.Dalton’s atomic theory is based on two other important chemical laws of the time the Law of conservation of mass by Antoine Lavoisier and the Law of definite proportions by Joseph Proust.The atomic theory was developed by John Daltons after he made the observation that chemical substances combine and break down into other substances by weight which suggested that such chemical substances are made of smaller particles with a particular weight.Dalton’s atomic theory is a scientific concept which states that all matters are composed of tiny indivisible and discrete particles called atoms.Dalton introduced atomic theory via a series of articles called ‘A New System of Chemical Philosophy’ which was published in 1803.His work on color blindness was also quite popular as Daltonism became a common term for color blindness. Besides the atomic theory, John Dalton also introduced other concepts in chemistry like gas laws.John Dalton has made numerous contributions to different areas of science, but one of his most important contributions is the introduction of the atomic theory in chemistry.John Dalton (6 September 1766- 27 July 1844) is an English meteorologist, teacher, and chemist who postulated the atomic theory and is considered a pioneer in modern chemistry. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
